Taking Piroplasmosis Seriously
- Topics: Article, Basic Care, Biosecurity, Horse Care, Insect Control, Piroplasmosis
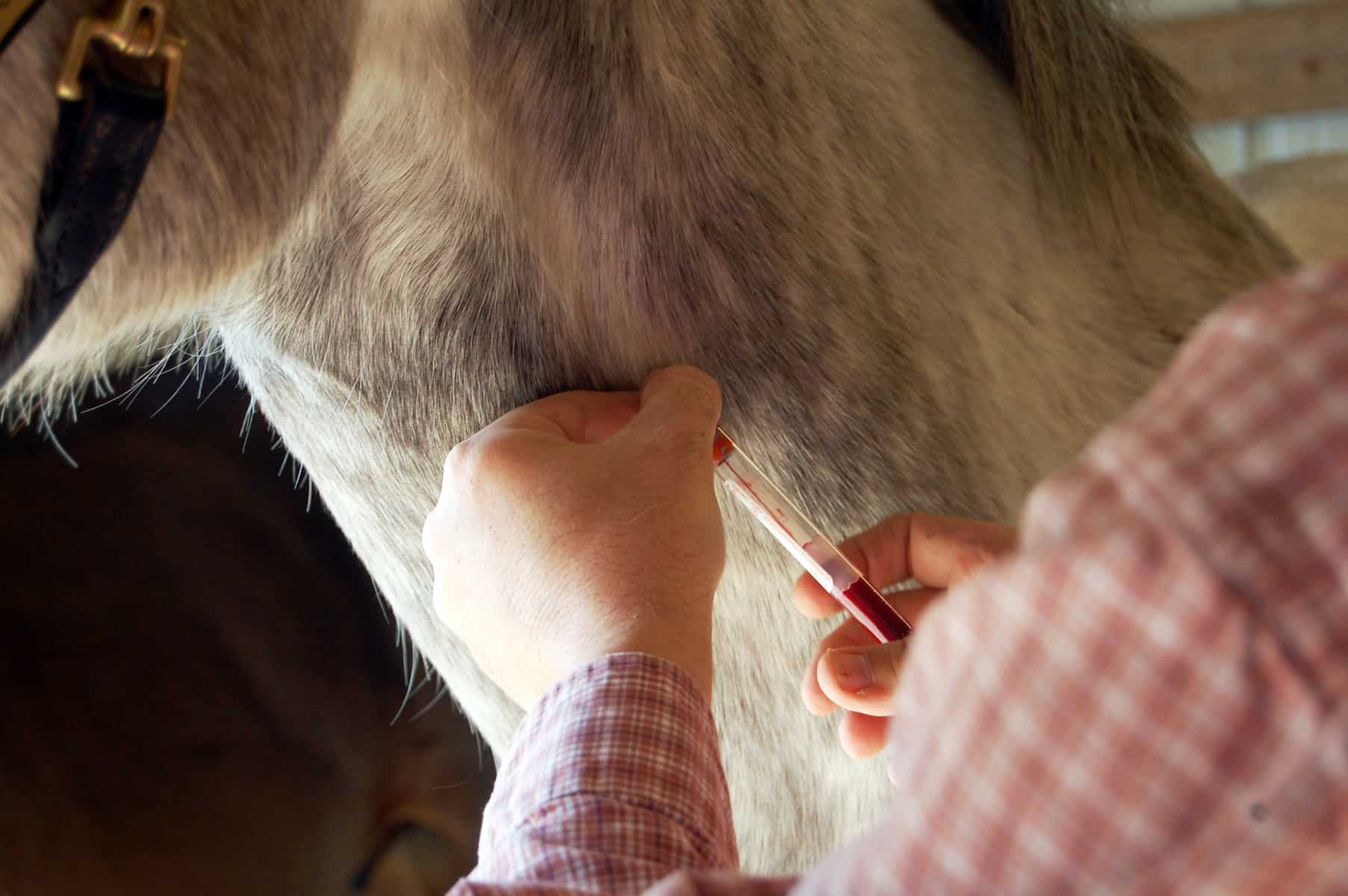
To prevent this parasitic disease from taking hold in the United States, veterinarians are fine-tuning testing, prevention, and treatment methods
In most parts of the world, equine piroplasmosis (EP) is endemic, routine, even commonplace. In the United States, however, this foreign animal disease is reportable and subject to regulatory action to mitigate disease spread. It is a parasitic disease of all equids, caused by Theileria equi and/or Babesia caballi. In endemic (native) countries ticks or other arthropods transmit EP from one infected horse to another. However, people can spread disease agents as well, by reusing needles or syringes or using blood-contaminated instruments such as dental or surgical equipment that have not been cleaned and disinfected appropriately between uses.
Pathogens and Pathology
Upon introduction to the horse’s body, both T. equi and B. caballi invade the horse’s red blood cells and can cause severe anemia (low red blood cell count) in some infected animals, evidenced by lethargy, reduced performance, and pale mucous membranes. Horses might also develop fever, icterus (jaundice), anorexia, and digestive problems including colic, constipation, or diarrhea. And because these clinical signs can also accompany other diseases, pursuing proper diagnostic testing is crucial. Veterinarians can detect EP by identifying the parasites on blood smears in clinically ill horses or through serologic testing using the competitive enzyme-linked immunosorbent assay (cELISA) or complement fixation (CF) tests. A third type of serology test, the immunofluorescent antibody (IFA) test, is also available to veterinarians, and scientists have developed polymerase chain reaction (PCR) tests for research purposes.
Picking Up on EP
In EP-endemic countries most horses become infected within their first year of life, and their fatality rate (5-10% of affected horses) is significantly lower than that in naive horses transported to endemic areas (which can exceed 50%) TheHorse.com is home to thousands of free articles about horse health care. In order to access some of our exclusive free content, you must be signed into TheHorse.com. Already have an account?Create a free account with TheHorse.com to view this content.
Start your free account today!
and continue reading.
Written by:
Christy Corp-Minamiji, DVM
Related Articles
Stay on top of the most recent Horse Health news with















